Hugo Lino Cavallone and Carlos R. Salvarezza
Acute respiratory failure (ARF) is a complex entity, which develops on the basis of numerous pathophysiological mechanisms and which frequently endangers the lives of patients.
Respiration is the movement of gas molecules through permeable membranes.
There are two stages in respiration: a) external or pulmonary, through which an organism exchanges gases with its environment, and b) internal or cellular, where the uptake and use of oxygen and the elimination of carbon dioxide through the tissues takes place. .
ARF occurs when the respiratory system is unable to meet the metabolic needs of the body. This implies that the essential condition for the diagnosis of ARF is the presence of hypoxia (insufficient supply of oxygen to the tissues). As there are no specific clinical or laboratory elements to determine when there is hypoxia, ARF is defined by the degree of hypoxemia (decrease in arterial oxygen pressure), from which it is inferred that there is also hypoxia.
A patient is in ARF when the arterial oxygen pressure (PaO2) is less than 50 mm Hg, with or without an increase in the arterial pressure of carbon dioxide (PaCO2), breathing room air and in the absence of a right-to-left intracardiac shunt .
IRA is classified into two fundamental types:
Type I: when there is hypoxemia with normo or hypocapnia (hypoxemic failure, not ventilator).
Type II: when there is hypoxemia and hypercapnia (ventilatory failure).
 Pathophysiology
Pathophysiology
The respiratory centers are located in the brain stem and in the cerebral cortex.
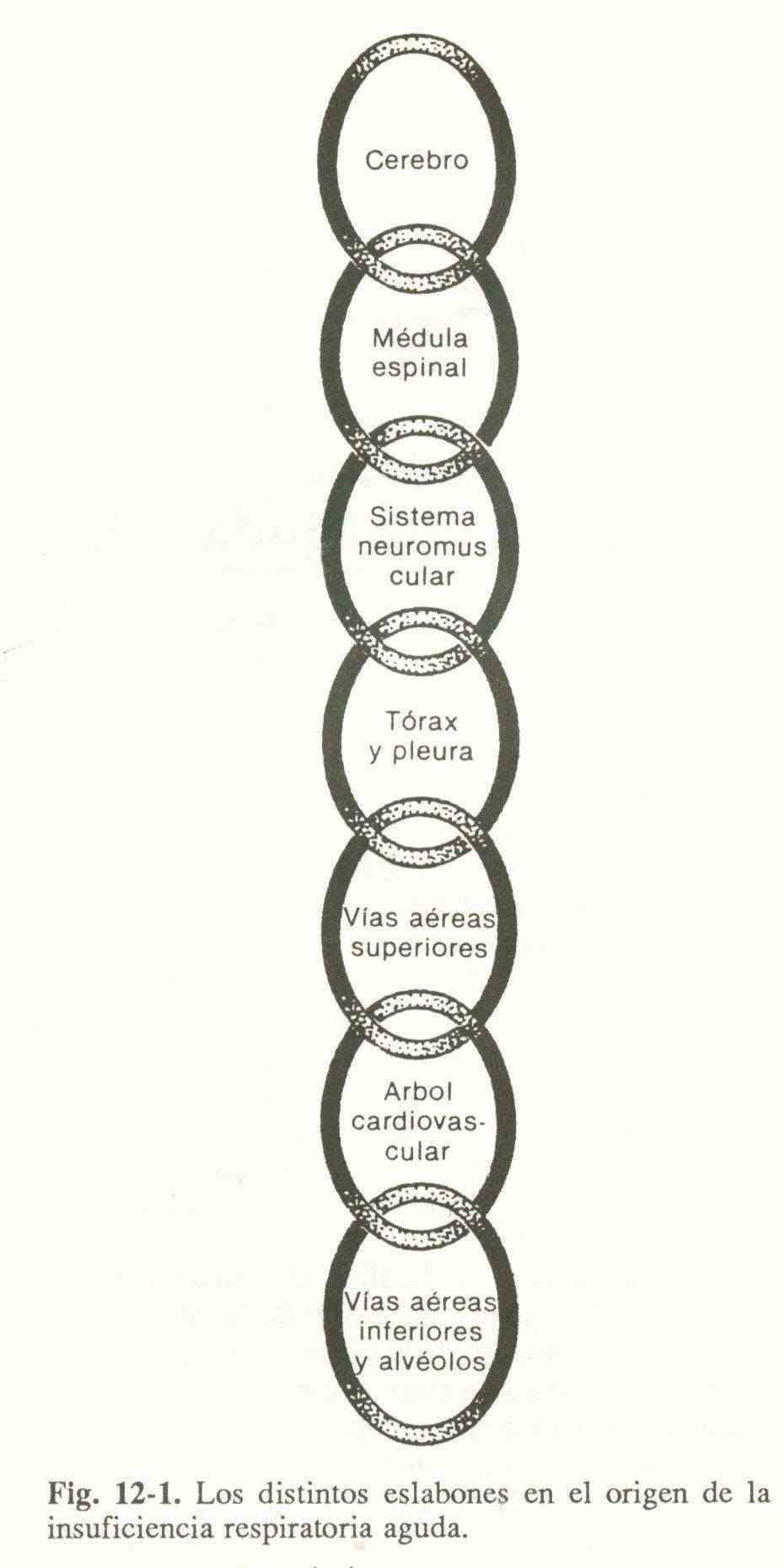
During the process of respiration, an exchange of gases takes place between the environment and the cells. For your better understanding, four functional subdivisions are considered:
|
|
Hypoxia develops by the following pathophysiological mechanisms:
|
The integrating neurons of the spinal cord process the efferences of both respiratory centers with the afferents of the peripheral proprioceptors and send signals through the phrenic and intercostal nerves to the neuromuscular plate, causing the expansion of the thoracic cage and generating a negative intrapleural pressure. that makes the gaseous mixture move through the airway to the alveoli. This can be schematized (Figure 12-1) by a series of links that extend from the brain to the alveoli. The alteration of one of these rings can cause an ARF.
Table 12-1 lists the most frequent causes that affect each ring in the chain.
a.Hypoxemic hypoxia.
1) decrease in FiO2. it happens when you breathe high above sea level. The decrease in FiO2 directly modifies PaO2.
2) alveolar hypoventilation. It is the minute ventilation that cannot maintain a normal PaCO2 for a certain metabolic state. Taking into account the equation for alveolar gases (which are described in the diagnostic methodology apparatus), any increase in PaCO2 is accompanied by a decrease in alveolar oxygen pressure (PAO2) and therefore PaO2. This mechanism is present in patients with healthy lungs who have cerebral, neuromuscular or skeletal alterations, or depression due to sedatives or anesthetics.
3) alteration of diffusion. It occurs when there is the ability to balance gases on both sides of the alveolocapillary membrane. Capillary blood normally balances the alveolar oxygen tension in the third part of the time that it is exposed for its exchange. For hypoxemia to develop through this mechanism, there must be a gross alteration of the alveolocapillary membrane, as can occur in fibrosis or pulmonary edema, and a shortening of the circulation time (exercise, tachycardia).
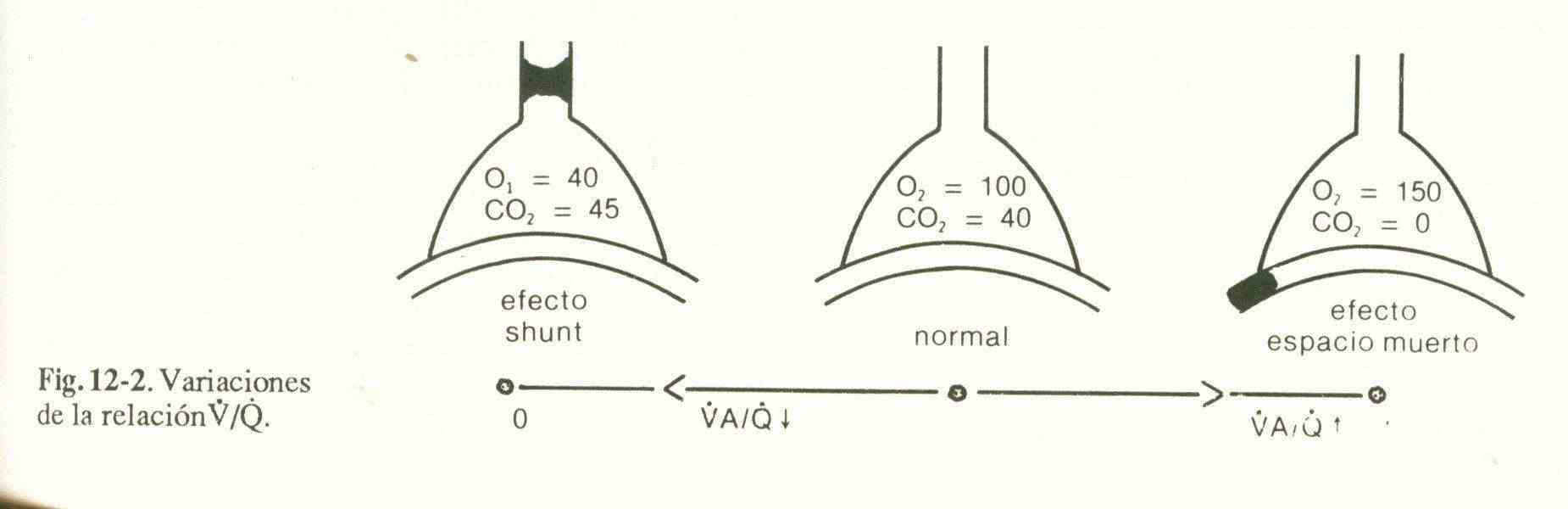
4) Ventilation / perfusion inequality (V / Q). For effective gas exchange there must be a certain adjustment between ventilation and perfusion. Possible variations in this relationship are shown in Figure 12-2.
Well ventilated, non-prefused units can only cause hypercapnia when a large area of the lung is involved.
Units that have a good V / Q ratio and those that are not ventilated or pre-fused do not generate changes in arterial blood gases.
5) Intrapulmonary shunts. They are an extreme form of V / Q irregularity that occurs when there are alveolar units with good perfusion but without ventilation (V / Q = 0). Hypoxemia is not corrected with 100% oxygen therapy, which differentiates it from the other imbalances between V and Q.
There is an anatomical shunt consisting of the bronchial arteries, the blood of which reaches the left atrium through the pulmonary veins, and the coronary arteries, which flow into the left ventricle through asveins of Thebesio.
The anatomical shunt is less than 3% of the cardiac minute volume.
6) Decreased oxygen pressure in mixed venous blood (PvO2). The blood that returns to the heart has different oxygen pressures because they come from different vascular territories that supply organs with varied metabolic activity. This blood is mixed in the right atrium and ventricle, so it is in the pulmonary artery that the samples taken are representative of mixed venous blood.
When cardiac minute volume falls or tissue metabolism increases, PvO2 decreases and, if oxygen pressures cannot be balanced during alveolar-capillary exchange, PaO2 will also decrease. Hypoxemia is more important when the PvO2 is lower, when it is associated with ventilation-perfusion irregularities, and when the alveolocapillary exchange time is shorter.
b) circulatory hypoxia.
The supply of oxygen to the tissues depends on the cardiac minute volume and the oxygen content in the arterial blood. A sudden and important decrease in cardiac output directly alters the oxygen supply to the cells.
c) Anemic hypoxia.
The arterial oxygen content is determined by the concentration of hemoglobin (Hb), the capacity to transport oxygen (1.39 ml of O2 / g of Hb) and the degree of saturation of Hb.
A decrease in a Hb concentration below 8-9 g / dl can cause hypoxia, especially if it is not compensated by an increase in cardiac output.
The affinity of Hb for oxygen depends on plasma pH, PaCO2, intraerythrocyte 2-3-diphosphoglycerate concentration, and body temperature. The decrease in pH and the increase in the other factors, deviates the Hb dissociation curve to the right, decreasing its affinity for oxygen and favoring its release into the tissues. The reverse situation makes peripheral oxygenation difficult.
Carbon monoxide poisoning produces a shift to the left of the dissociation curve and a drop in the Hb saturation percentage, thus deteriorating the oxygen supply to the tissues.
d) Dissoxic hypoxia.
Dysoxia is the abnormal use of oxygen by cells. In some mitochondrial metabolic disorders, in cyanide intoxication and in septic shock, the use of oxygen is inadequate even if its supply is normal or increasing.
| Table 12-1. Causes of acute respiratory failure | |
|
|
 Symptoms and signs
Symptoms and signs
The clinical manifestations of ARF are very varied and this is due to the multiplicity of causes that originate it.
The first symptoms are disorientation, restlessness, irritability and depression of intellectual functions. Patients generally report dyspnea and may be tachypneic, distressed, and short of breath. Cyanosis of the skin and mucosa is usually present, denoting an increase in reduced Hb. Other symptoms are tachycardia, peripheral vasoconstriction, arterial hypertension and finally bradycardia and hypotension.
The earliest clinical findings associated with hypercapnia are throbbing headache and mental confusion, and are due to vasodilation and cerebral edema.
Other manifestations are asterixis, myoclonus, peripheral vasodilation, arrhythmias, and profuse sweating. As the condition enlarges, intracranial hypertension, seizures, arterial hypotension, mydriasis, respiratory depression, and coma appear.
Study methodology
Interrogation and physical examination.
The first symptoms of an ARF are due to cerebral hypoxia, but they are not specific because they can lead to toxic lesions or CNS infections. However, the anamnesis and physical examination allow us to orient ourselves towards the diagnosis of ARF, which will be confirmed with complementary studies.
Complementary studies
The goal of these tests is to find a parameter that measures gas exchange at the tissue level, because oxygen utilization can be good with a low PaO2 and bad with a normal or high PaO2.
a) lactic acid. Lactic acidosis is possibly the most certain manifestation of inadequate tissue oxygenation, although its presentation is usually very late.
b) PvO2. Until a few years ago, this parameter was considered an indicator of tissue oxygenation. It is currently suggested that a patient with a PvO2 less than 30 mm Hg (normal 40-50 mm Hg) has a high probability of being hypoxic, but being normal or high does not always mean adequate oxygenation. Added to this fact is the difficulty of extracting blood for its determination due to the need to place a catheter in the pulmonary artery.
c) Acid-base state and gasometry. To diagnose an ARF and evaluate its severity and response to oxygen therapy, the acid-base status and blood gas obtained by puncturing a peripheral artery are used.
In hypoxemic ARF (type I) there is also a decrease in PaCO2 with an increase in pH (respiratory alkalosis).
In type II ARF, in addition to hypoxemia, there is an increase in PaCO2 and a decrease in pH (respiratory acidosis).
d) alveolar-arterial oxygen difference (AaDO2). Assess the efficiency of gas exchange. The calculation of the PAO2 is established by applying the alveolar gas equation:
PAO2 = FIO x (PB-PH2O) - (PACO2 / R)
Where FiO2 = inspired fraction of O2 (0.21 for ambient air); PB = barometric pressure (760 mmHg at sea level); PH2O = water vapor pressure (47mm Hg), and R = respiratory quotient (0.8).
Aa DO2 is obtained by subtracting PaO2 from PAO2. increases when oxygen transfer disorders occur; it has the advantage, with respect to blood gas measurement, that it is less sensitive to changes in ventilation, and the disadvantage that, under stable conditions, increases in Fio2 produce a widening of the gradient.
When pulmonary involvement is severe and it is necessary to use a high FiO2 to assess oxygenation, it is advisable to use the PaO2 / FiO2 ratio; its normal value is 500, being an index of severity when it is less than 250; in turn, the PaO2 / PAO2 (a / A) ratio, which is normally 0.8, in ARF is less than 0.5.
Radiology . The chest radiograph is an important auxiliary to the clinician. It can approximate the etiological diagnosis, and detect complications that occur during the course of ARF and / or its treatment.
It is usually normal when the cause is in the CNS or neuromuscular plate, or present pathological images in case of pleural effusion, rib fracture, foreign bodies in the airway, pulmonary edema, pneumothorax, pneumomediastinum, pneumonia, atelectasis or diaphragmatic ascent , or evidence of previous cardiopulmonary diseases.
Other methods . With different results, other methods such as intravascular electrodes and transcutaneous measurement of blood gases have been used for the diagnosis or follow-up of patients with ARF.
The improvement of oxygen monitoring with sensors inserted directly into the tissues will probably be of great use in the future.